CH 610A/618A
First Exam
Fall, 2004
Prof.N.L.Bauld
__________________________________________ Name (Last,
First)
CH 610A/618A
First Exam
Fall, 2004
Prof.N.L.Bauld
__________________________________________
Name
(Last, First)
Question |
Possible Points |
Page/Pts/Points Earned |
Score |
|
I. |
27 |
3/07/ 4/08/ 5/08/ 6/04/ |
|
|
II. |
26 |
7/07/ 8/11/ 9/8/ |
|
|
III. |
22 |
10/7/ 11/06 12/09 |
|
|
IV. |
12 |
13/9/ 14/3/ |
|
|
V. |
13 |
15/08/ 16/05/ |
|
|
Totals |
100 |
|
|
I. Ground State Electronic Configurations and Hybridizations
A. Electronic Configurations.
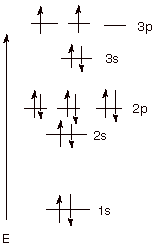
1. [3 pts] Write the ground state electronic configuration
of positively charged phosphorus (15P + ; phosphorus is in Group V of the Periodic
Table) in the energy level diagram
format.
2.
[4 pts] Name the three
rules which are needed to derive the correct ground state configuration and
explain how they are applied to the case of positively charged phosphorus.
a. The Aufbau Rule: Fill in electrons beginning with
the lowest energy orbital first and proceeding to orbitals of progressively
higher energy.In the above case, electrons go into the 1s orbital first, then
the 2s, then the 2p orbital.
b. The Pauli Exclusion Principle: A maximum of two
electrons can occupy any orbital but their spins must be paired. Two electrons
can go into the 1s and 2s orbitals, if their spins are paired.
c. The Hund Rule: In the case of degenerate
(equienergetic) orbitals, the lowest energy state is provided by filling
electrons into different orbitals rather than into the same orbital, and with
their spins unpaired, i.e., having the same spin. The fifth and sixth electrons
then go into different 2p AO’s, but they have the same spin.
B. Electronic Configuration and Hybridization of Carbon.
1. [4 pts] Using the linear format only, write the ground state electronic configuration
of (neutral) carbon atoms (6C ) and show in stepwise fashion (two
steps) how carbon atoms hybridize when they are bound to four other atoms or
groups, as in CH4.
Be sure to include (1) the names of any other states involved in
addition to the ground state (2) the potential valence of carbon in that state
and (3) comment on the energetic feasibility of each of the individual steps and of the overall process of hybridization to relevant
final state.

The promotion of a 2s
electron to a 2pz orbital requires about 96 kcal/mole of energy (ca.
100 kcal/mol is close enough). The increase in valency from 2 to 4, provides
two extra bonds at ca. 100 kcal/mol apiece. Consequently, the increased number
of bonds more than supplies the need for energy to promote the electron.
2. [2 pts] Specify two charged elements which have the same electronic configuration as the neutral atomic carbon discussed above? One of these should be positively charged and the other negatively charged. Why do these charged elements have exactly the same electronic configuration as neutral carbons?
q
Positively charged nitrogen
q
Negatively charged boron
q
Because they have the same number of electrons as
neutral carbon.
3.
[2 pts] What is the name
of the principle which
interrelates neutral atomic carbon and these charged atoms? Predict the
geometries of the structures which are formed when each of the two charged
atoms of the previous question are bonded to four hydrogen atoms. Give reasons
for your answers.
q
The Isoelectronic
Principle
q
Tetrahedral, the same
as methane.
4.
A. [4 pts] Draw an energy
level diagram which shows the
energies of the 2s and 2p atomic orbitals of atomic carbon. Then place the
energies of the sp, sp2, and sp3 hybrid orbitals on the same diagram, using
care to display the energies of these latter orbitals not only in the right
energy sequence but with approximately the right spacings between these hybrid
orbitals and in relationship to the 2s and 2p AO energies.
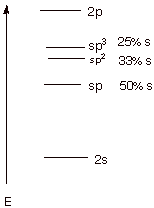
5. [2 pts] What percentage
of s character is in each of these
three hybrid AO’s? Indicate how this percentage is derived.
q Percentages shown in above diagram.
q For example, the sp3 AO is one part s
and three parts p; therefore it is 1/4 or 25% s.
6. [2 pts] Qualitatively, compare the bond lengths of the C-H
bonds formed when each of the three hybridization states of carbon bonds to
hydrogen. Explain why the bond lengths are different and why the sequence of
lengths is that observed.
q
The C-H bond lengths
decrease as the % s character increases (e.g., shortest in the sp AO and
longest in the sp3 case.
q
This is because the stronger a bond is, the shorter
it is. The sp bond is stronger because the electrons are in an MO which is
comprised of a low energy sp AO, overlapping with a H 1s AO.
C. Alkenes
1. [3 pts] Starting with the ground state electronic
configuration of neutral carbon, sketch the stepwise development of the
hybridization state of carbon in ethylene (ethene) and other alkenes. What is
the designation of this hybridization state?
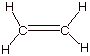

q
Designation is
trigonal.
2. [1
pt] When (under what general circumstances) does
neutral carbon adopt this hybridization state?
q When it bonds to (just) three other atoms. (Examples would include carbocations and radicals as well as alkenes and aromatics).
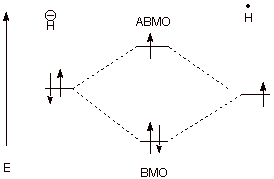
II. Atomic Orbitals, Molecular Orbitals, and Bonding
A. Carbon-Carbon Bond Formation
1.
The overlap of two carbon 2p AO’s in the manner shown below can result in
the formation of a strong covalent bond.
a. [3 pts] Referring to the diagram given below, label the
two new orbitals which are formed when these two atomic orbitals overlap,
providing both the abbreviations and the full names of the orbitals. Explain
the specific basis for the names assigned to them.
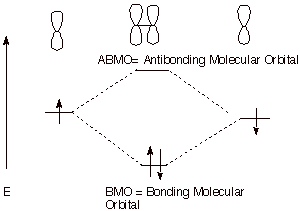
The reference point is the energy of the starting
atomic orbital. An MO of energy more negative than this is bonding; an MO
having an energy less negative than this (higher in energy) is antibonding.
b. [2 pts] Explain why the two electrons both go into the
same orbital. According to what principle must their spins be paired? What
unallowable circumstance would necessarily result if (somehow) the third
electron were added (not just a violation of the rule above).
q
Both pi electrons
enter the same orbital (the BMO) because this is by far the lowest energy
orbital available (in other words, it is the Aufbau principle).
q
Their spins must be
paired as a consequence of the Pauli Exclusion Principle.
q
If a third electron
were added to the same orbital, it would have to have the same spin as one of
the two electrons already present. This would make two indistinguishable
electrons, a situation precluded by the Pauli Exclusion Principle.
c. [2 pts] Given the geometric nature of the overlap
depicted in the diagram above, what kind of covalent bond is this? What kind of
overlap is involved in this kind of bond?
q
A Pi bond.
q
Sidelong or lateral
overlap.
d. [3 pts] Provide a depiction of both of the new orbitals
which are formed in the bond formation process indicated above in terms of
their constituent 2p orbitals. Include the signs of the lobes and any nodal
planes. In terms of a specific type of overlap, what is different about the low energy orbital and
the high energy orbital?
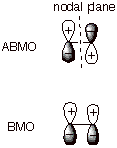
q
The ABMO has
“negative overlap”, i.e., overlap between lobes of opposite sign.
e. [2 pts] Compare the
relative efficiency of the type of overlap shown above and found in ethene,
with the type which is found in ethane. What is the name of the type of overlap
found in ethane?
q
The overlap in ethane
is sigma type or end-on overlap.
q
Sigma overlap is more
efficient than pi overlap, so the sigma bond is stronger.
2.
A bond line structure for ethene is given below. The following questions refer
to the bonding in ethene.
a.
[2 pts] Provide an orbital depiction of all of the sigma bonds in ethene (carbon-hydrogen and carbon-carbon).
b.
[4 pts] Explain why all six atoms lie in the same plane and why rotation around
this bond involves breaking a bond. Use Newman projection structures to depict
your answer.
q
Both carbons are sp2
hybridized and individually coplanar with the three attached groups (H,H, and
C).
q
The trigonal planes
of the two trigonal carbons are additionally required to be coplanar because only in that way is efficient pi overlap
effected.
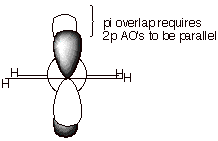
B. Strong and Weak
Covalent Bonding.
1. [3 pts] Specify the general requirements for strong covalent bonding, briefly explaining the basis for
each requirement.
q
Efficient (or
strong), net overlap.
q
Two paired electrons.
q
Since only one
bonding MO is formed it can accommodate a maximum of 2 electrons, and then only
if there spins are paired (Pauli Exclusion). Bonding is maximized if this low
energy orbital is fully occupied (2 e).
q
In general, the
stronger the overlap, the lower the energy of the BMO, so strong overlap gives
more bonding. Net overlap refers to the possibility that there can be strong
overlap in one region, but negative overlap in another, and the two may cancel,
yielding no net overlap.
2. [3 pts] Cite two specific situations in which atoms closely approach and efficient
orbital overlap results, but no
covalent bonding at all results. The situations should be fundamentally
different. Provide energy level diagrams illustrating each of these cases.
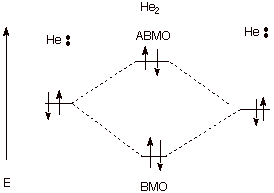
q
In the case of the close
approach of two helium atoms, strong overlap occurs and BMO/ABMO formation
occurs, but there are four electrons (each He has two electrons), so that the
amount of antibonding which ensues from the ABMO cancels the bonding which
would have been accrued through the BMO.
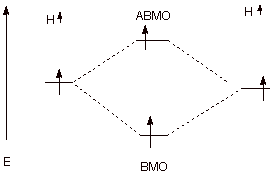
In the case of close
approach of two hydrogen atoms which both have the same spin (spins unpaired),
there are two electrons, but they cannot be put into the BMO. One must, by the
Pauli Principle, occupy the ABMO, again negating the bonding effect of the BMO.
3. [2 pts] Cite two specific situations in which atoms closely approach and efficient
orbital overlap occurs, but only weak or modest covalent bonding results. Again, the two situations
should be fundamentally different. Provide energy level diagrams illustrating each of these cases.
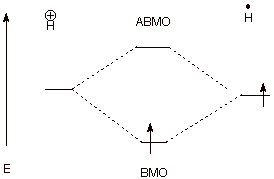
q
The close approach of
a proton to a hydrogen atom leads to strong overlap and BMO/ABMO formation, but
there is only one electron to place in the BMO, so that net, but moderate,
bonding occurs.
q
In this case, a
hydride anion (which has a hydrogen nucleus with two electrons) approaches a
hydrogen atom. There is weak, net bonding because there are two electrons in
the BMO and only one in the ABMO, so there is not full cancellation of the
bonding effect.
III. Nomenclature, Formal Charges, and Resonance Structures
A. Formal Charges.
1. [5 pts] Calculate the formal charges on each atom other than hydrogen in the following
structures. Provide the formula you are using, and show your calculations.
Then, calculate the net charge on
the species. The atomic number of oxygen is 8 and that of nitrogen is 7.
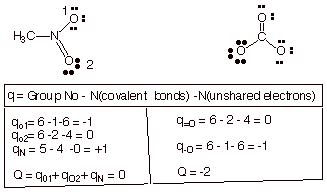
B. Resonance Structures.
1. [2 pt] Indicate what kind of P-C bonding (other than the
sigma bond) is involved in each of the two resonance structures below. Which
structure is the lower energy one? Why?
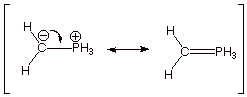
q Ionic bonding in the first structure.
q Covalent, pi bonding is involved in the second.
q The first is considered to be of lower energy in this case because phosphorus has to use a very high energy 3d orbital to overlap with the carbon orbital (it already has used all four of its sp3 AO’s to form sigma bonds.)
2. [2 pts] Using the appropriate formalism of resonance
theory, describe the acetate ion (one
canonical structure is shown below) in terms of resonance theory.
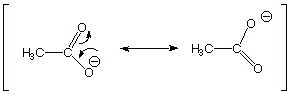
3. [1 pt] Summarize this as a dotted line/partial charge
structure.
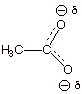
4. [1 pt] The resonance energy of the acetate anion is
characterized as large. Upon what
basic principle of resonance theory is this based?
q
Resonance
stabilization is particularly large when the resonance structures are equivalent in energy.
5. [1 pt] What is the main geometric consequence of resonance in the acetate anion?
q
The two C-O bonds are
equal in length and intermediate between single and double.
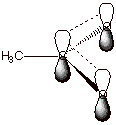
6. [1 pt] Provide an orbital depiction of the pi electron
system of the acetate anion. Such an extended (more than two AO’s are
involved) pi electron system is termed a conjugated system. Over how many atomic orbitals are the
MO’s of this conjugated system delocalized?
q
Delocalization of the
electrons is over three atoms, the two oxygens and the carbonyl carbon.
C.
Nomenclature
1. [3 pts each = 6 pts]
Provide complete, accurate IUPAC names for the following molecules. As a part
of your answer, include the numbering system and the basis for your choice of
numbering system.
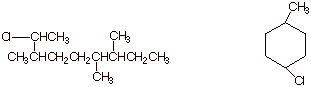
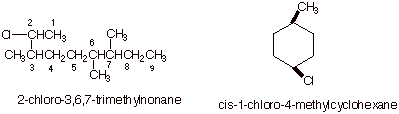
3. [3 pts] Provide the IUPAC name for the following
molecule, using the appropriate IUPAC substituent name.
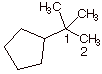
q
1,1-dimethylethylcyclopentane
IV. Conformational
Analysis; Torsional Strain; Steric Effects.
A. Conformational Analysis of Butane.
1. [5 pts] Sketch an energy vs. dihedral angle conformational energy plot for butane, providing the names, Newman projection structures, and relative energies of all conformational extrema.
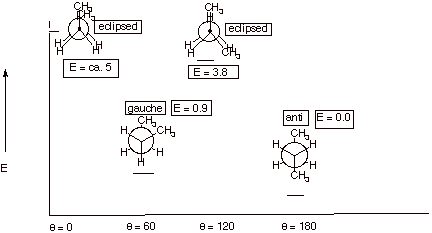
2. [2 pt] What is the name of the type of effect which
accounts for the difference in energies of the two energy minima of butane.
Describe the fundamental basis for this effect and provide a depiction of it.
q
Steric effect or van der Waals repulsion.
q
A repulsion between hydrogen atoms of the methyl
groups which are too close, i.e., closer than the sum of the van der Waals
radii.
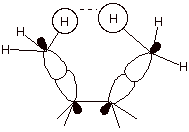
3. [2 pts] Explain the difference between torsional
strain and steric strain , using a depiction to assist in illustrating your
answer.
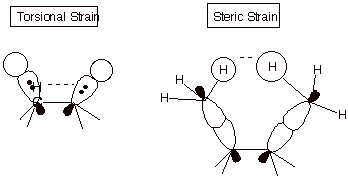
q
Steric strain derives from a repulsion between
atoms which are too close, while
torsional strain derives from the repulsions of electron pairs in bonds, which are closer in the eclipsed than in the
staggered conformation.
C. Ring Strain.
1. [3 pts] Using the diagram provided below as a starting
point for your illustration, describe and illustrate the basis for the angle
strain present in cyclopropane. Be certain to include the concepts of
interorbital angle and internuclear angle in your explanation.
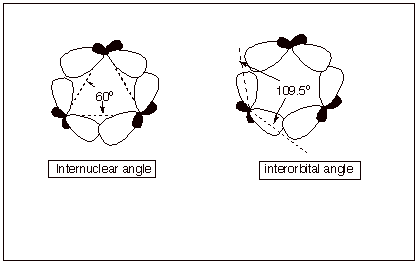
q
The internuclear angle for an equilaterial triangle
is 60 degrees, but the interorbital angle for sp3 orbitals
on the same atom is the tetrahedral angle.
q The result of this non-correspondence is that the orbitals on the ring carbons can not point toward each other, as is the case for a typical, strong carbon-carbon sigma bond. The bond is “bent”.
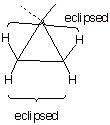
2. [2 pts] What other type of strain is present in
cyclopropane? Explain the basis for this strain, and indicate which bonds are
responsible for it.
q
Cyclopropane also has torsional strain because, being planar, all of the C-H bonds are eclipsed.
V. Conformations of
Cyclohexane and Substituted Cyclohexanes.
A. Cyclohexane Conformational analysis.
1. [2 pts] Draw the conformational structure of the conformation of cyclohexane which represents the energy minimum. Show clearly on your structure and label the axial and equatorial carbon-hydrogen bonds (all of them).
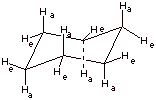
2. [3 pts] How much strain is present in this conformation of cyclohexane? Explain your answer in detail, with reference to both types of strain which are typically present in cycloalkanes.
q
Zero strain.
q
There is no torsional strain because all of the C-H
and C-C bonds are staggered.
q
There is no angle strain because all of the C-C-C
angles are 109.5 degrees.
3. [1 pt] Draw the structure of this energy minimum conformation again and then show (connected by a equilibrium arrows) the structure which would result upon “ring flip”. (Do not show the detailed conformational path at this point, but just the final ring flip structure.
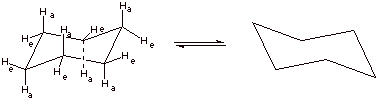
4. [2 pts] Draw and name the structure of the global energy maximum on the ring flip path, and indicate its energy relative to the minimum energy structure. What is the importance of this structure?

q
This is the “half chair” conformation.
q
Its energy is 10 kcal/mol greater than that of the
chair
q
The importance is that this is the highest point on
the ring flip path and determines the activation energy and therefore the rate
of the flip.
B. Monosubstituted Cyclohexanes.
1. [3 pts] Draw the conformational structure of the conformation of methylcyclohexane which is of lowest energy and show the structure which results upon ring flip. Provide the names and relative energies of these two structures.
2. [3 pts] Draw the conformational structure of the conformation of methylcyclohexane which is of lowest energy and show the structure which results upon ring flip. Provide the names and relative energies of these two structures.
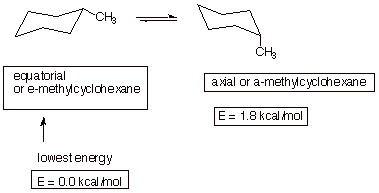
2. [2 pts] Using a depiction to assist in clarifying your answer, explain which structure is the global minimum, what type of strain is present in the less stable conformation, and the basis for the strain.
q
As noted, the equatorial conformation is more
stable.
q
The axial conformation has steric strain or diaxial
strain. Alternatively, it can be described as the equivalent of two gauche
butane interactions.
q
This arises from the hydrogens of the methyl group
repulsing each of the other two axial hydrogens on the same same of the
generalized plane.